WuXi AppTec Reports Record First-Quarter 2021 Results
2021/04/29
Revenue Up 55.3% Year-Over-Year to RMB4.95 Billion
Net Profit Attributable to Owners of the Company Up 394.9% Year-over-Year to RMB1.50 Billion
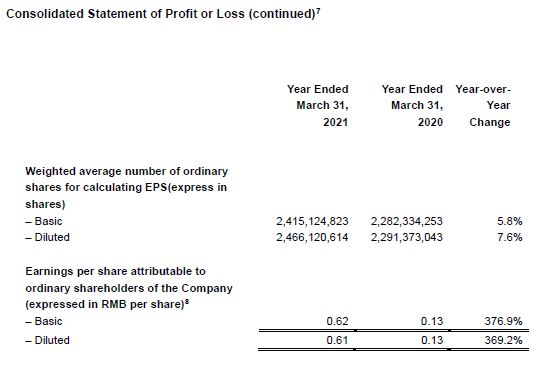
Diluted EPS Up 369.2% Year-Over-Year to RMB0.61
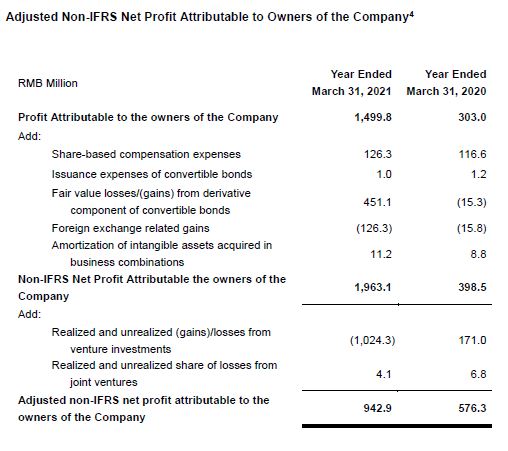
Adjusted Non-IFRS Net Profit Attributable to Owners of the Company Up 63.6% Year-Over-Year to RMB943 Million
Adjusted Diluted Non-IFRS EPS Up 52.0% Year-Over-Year to RMB0.38[1]
[1] In the three months ended March 31, 2021 and three months ended March 31, 2020, we had a fully-diluted weighted average share count of 2,466,120,614 and 2,291,373,043 ordinary shares, respectively
First-Quarter 2021 Financial Highlights
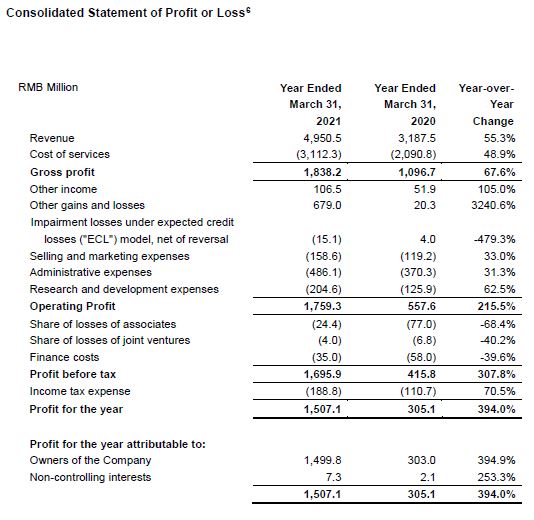
- Revenue soared 55.3% year-over-year to RMB4.95 billion on the back of robust demand brought on by strengthened customer market penetration and accelerated customer expansion. Revenue up 4.9% quarter-over-quarter continued our strong quarterly-based growth track record in the past twelve quarters. China-based laboratories and contract development and manufacturing organization (CDMO) facilities maintained high utilization to meet customer demands. Clinical research services experienced a strong rebound, but headwinds remain strong for US-based laboratory services which has continued to be negatively impacted by the COVID-19 pandemic.
·China-based laboratory services revenue grew 49.0% to RMB2.56 billion.
·CDMO services revenue grew 100.0% to RMB1.70 billion.
·U.S.-based laboratory services declined 15.3% to RMB329 million.
·Clinical research and other contract research organization (CRO) services revenue grew 56.8% to RMB359 million.
- IFRS gross profit increased 67.6% year-over-year to RMB1.84 billion. Gross profit margin was 37.1% vs 34.4% in first-quarter 2020.[2]
- Adjusted Non-IFRS gross profit increased 58.3% year-over-year to RMB1.94 billion. Adjusted Non-IFRS gross margin was 39.1% vs 38.4% in first-quarter 2020.
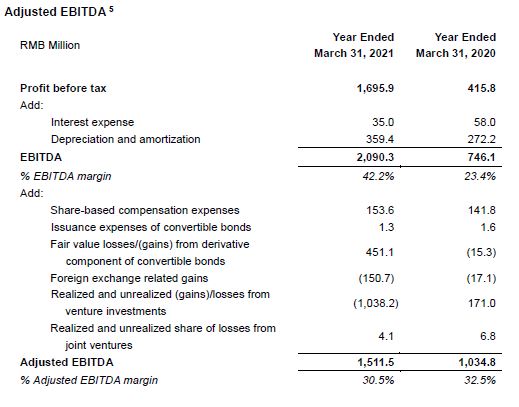
- EBITDA increased 180.2% year-over-year to RMB2.09 billion.
- Adjusted EBITDA increased 46.1% year-over-year to RMB1.51 billion.
- Net profit attributable to owners of the Company increased 394.9% year-over-year to RMB1.50 billion. Our gain from venture investment portfolios and loss from associates totally contributed a net gain of RMB1.02 billion, due primarily to the stock price increase of certain listed companies and some portfolio companies successfully went IPO during the Reporting Period that led to an increase in their fair market value and an RMB4 million loss from our joint ventures. Conversely, in the first-quarter 2020, we reported a RMB171 million fair value loss from investment portfolios and associates, and a RMB7 million loss from our joint ventures.
- Adjusted Non-IFRS net profit attributable to owners of the Company increased 63.6% year-over-year to RMB943 million.
- Diluted EPS increased 369.2% year-over-year to RMB0.61, while adjusted diluted non-IFRS EPS increased by 52.0% year-over-year to RMB0.38.
[2] If prepared under Accounting Standard for Business Enterprises of PRC, 2021 the gross profit grew 67.2% year-over-year to RMB1.84 billion. Gross profit margin was 37.1%.
First-Quarter 2021 Business Highlights
- We continued to relentlessly execute our strategy of enabling customers to innovate and accelerate drug discovery and development by leveraging our global integrated R&D services platform. We added over 360 new customers in the first-quarter of 2021, giving us a total of more than 4,400 active customers.
·Our global platform continued to enable innovation worldwide. During the Reporting Period, our overseas customers contributed RMB3.62 billion in revenues, increasing 49.5% year-over-year. Our China-based customers contributed RMB1.33 billion in revenues, increasing 73.9% year-over-year.
·We continued to expand our customer base and retain existing customers. During the Reporting Period, our existing customers contributed RMB4.75 billion in revenue, representing a year-over-year growth of 54.3%. Our newly added customers in the first-quarter of 2021 contributed RMB197 million in revenue.
We aim to simultaneously increase service penetration in large global pharmaceutical companies, whilst increasing the size of our “long-tail” customer base. This strategy has continued to be successful. During the Reporting Period, the top 20 global pharmaceutical companies contributed RMB1.58 billion in revenue, increasing 66.6% year-over-year. Our “long-tail” and other customers contributed RMB3.37 billion in revenue, growing 50.5% year-over-year.
·We continued to increase customer conversion and deliver synergies across our entire platform. During the Reporting Period, customers using services from more than one of our business units contributed RMB3.94 billion in revenue, growing 58.4% year-over-year.
China-Based Laboratory Services: robust growth in all business lines on the back of strengthened customer market penetration and expansion
- Chemistry FFS (Fee for Services) achieved over 58% revenue growth while concurrently transferring multiple new projects to our CDMO segment.
- Through comprehensive integration of our DNA-encoded library (DEL), protein production and structure-based drug design capabilities, our Target-to-Hit platform(HitS) has enabled over 600 customers globally, and diverted multiple, incremental business opportunities to our downstream business units.
- As of March 31, 2021, our success-based drug discovery service unit had cumulatively submitted 120 IND filings with the National Medical Products Administration (NMPA) and obtained 91 Clinical Trial Applications (CTAs) and had two projects in Phase III clinical trials.
- Safety assessment / toxicology services revenue grew rapidly at approximately 114% due to strong demand and increased animal room capacity.
- We signed over 40 integrated WIND packages (the WuXi IND program or “WIND”) in the first-quarter of 2021.
CDMO Services: first-quarter 2021 growth doubled due to core business model execution and capacity increase
We added 169 new molecules into our small molecule CDMO pipeline, including 11 new projects that were transferred from clients’ facilities or other CDMOs. We provided CDMO services to over 1,340 active projects, including 46 projects in Phase III clinical trials and 28 projects in commercial manufacturing.
- In the first-quarter 2021, new construction at Taixing city has begun and will provide a large scale API and oligonucleotide and peptide API production once complete. Taixing site is designed to provide over 140,000 square meters of manufacturing space in 2022.
- The drug product manufacturing facility in Wuxi city slated to begin operation in 2021 will not only improve the development and production capacity of solid dosages, but will also be capable of sterile drug product development, clinical trial material production and commercial scale manufacturing.
- The high-potency API manufacturing facility, large-scale oligonucleotide API manufacturing facility and large-scale peptide API manufacturing facility located in Changzhou city began operations, supporting process R&D and small molecule manufacturing, as well as oligonucleotide and peptide APIs from preclinical to commercial.
US-based Laboratory Services: Continued development of a comprehensive US-based cell and gene therapy CTDMO platform, with the integration of OXGENE
Our cell and gene therapy Contract Testing Development and Manufacturing Organization (CTDMO) services enabled customers globally. During the Reporting Period:
·Our laboratories and facilities in the U.S. provided services for 36 clinical stage projects, including 22 projects in Phase I clinical trials and 14 projects in Phase II/III clinical trials.
·The current quarter revenue decline in our U.S. cell and gene therapy business was mainly due to delay in approval of commercial projects and impacted by the pandemic. Some late stage/commercial clients also did not pass clinical trials; however, we are building up our new projects pipeline through significantly enhanced viral vector platforms and through integration with the newly acquired new OXGENE platforms. We expect strong rebound in revenue growth in the second half of 2021.
·In our Medical Device Testing business, the impact of the pandemic continued first-quarter. The delay of elective/non-essential surgeries impacted key projects caused shortfall of the testing demand. We are actively working and supplementing with new opportunities, particularly the EU Medical Device Regulation (MDR) to grow the medical device testing business in the second half of 2021.
Clinical Research CRO/SMO Services: strong rebound in revenue driven by focused backlog execution and timely project delivery
- Our clinical research services continued to enable customers globally during the Reporting Period:
·Clinical development services (CDS) backlog increased approximately 56% on a year-over-year basis and our site management organization (SMO) backlog increased approximately 47% on a year-over-year basis.
·China based clinical research services[3] delivered strong growth in the first-quarter, at 64.7% year over year, while US based clinical trial services continued to suffer from the impact of the pandemic impact.
·CDS team provided services to more than 130 projects for our clients in China and the U.S. and completed registration trials for 3 products.
·Our SMO team maintained its No.1 leadership position in China, with more than 3,500 clinical research coordinators stationed in 150 cities providing services in ~1,000 hospitals. The team assisted in the market approval of 5 customer products that were approved by NMPA in the first quarter in 2021.
[3] China based clinical research services included CDS China and SMO businesses.
Management Comments
Dr. Ge Li, Chairman and CEO of WuXi AppTec, said, “Another record quarter has once again demonstrated the strength and resilience of our platform. We continue to flawlessly execute our business model, increasing customer penetration while increasing “long-tail” customer numbers with the highest quality of our service offerings. For CDMO, we are seeing benefits of aggressive investment in capacity and new modalities come to fruition with revenues doubling in the first-quarter. Our China-based laboratory services and clinical research services segments also out-performed, growing 49.0% and 56.8% respectively. We expect the upward trajectory of these business segments to continue, mitigating any continuing COVID-19 related challenges that U.S. based laboratory services may face.”
“A continued strong biotech funding environment, coupled with incremental demand from the pandemic, reinforces our decision to further invest in and expand our integrated platform solidifying our leading position to meet strong and fast growing demands in 2021 and beyond.”
Dr. Ge Li concluded, “This is a good start to 2021 and we anticipate this momentum to continue in the coming quarters. Going forward, we will continue to bolster our integrated business model by expanding our platform through investments in new modalities, further enabling our customers to bring the most innovative medicines to patients – fulfilling our vision that: ‘every drug can be made and every disease can be treated.’”
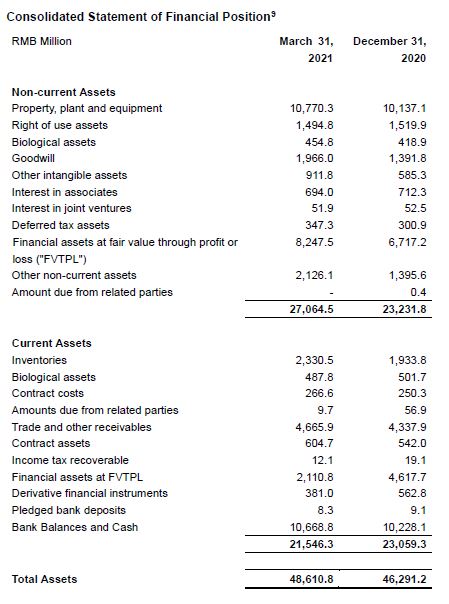
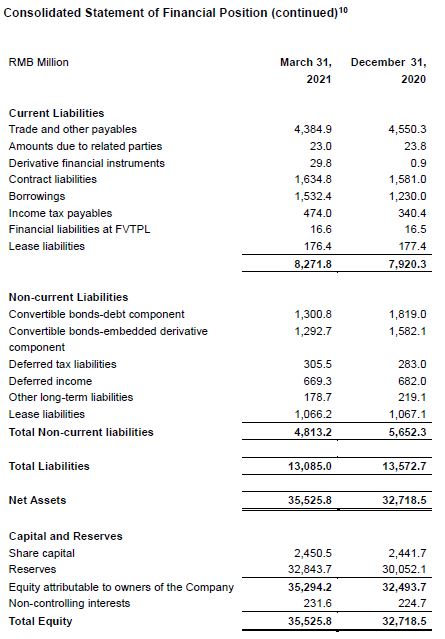
[4] If the sum of the data below is inconsistent with the total, it is caused by rounding.
[5] If the sum of the data below is inconsistent with the total, it is caused by rounding.
[6] If the sum of the data below is inconsistent with the total, it is caused by rounding.
[7] If the sum of the data below is inconsistent with the total, it is caused by rounding.
[8] In 2020, pursuant to the 2019 Profit Distribution Plan considered and approved by the shareholders’ general meeting, the Company issued 4 shares for every 10 shares of the Company by way of capitalization of reserve. In accordance with the regulations of the China Securities Regulatory Commission, the Company has adjusted the basic earnings per share and diluted earnings per share for the comparative period according to the 2019 Profit Distribution Plan.
[9] If the sum of the data below is inconsistent with the total, it is caused by rounding.
[10] If the sum of the data below is inconsistent with the total, it is caused by rounding.
About WuXi AppTec
WuXi AppTec provides a broad portfolio of R&D and manufacturing services that enable companies in the pharmaceutical, biotech and medical device industries worldwide to advance discoveries and deliver groundbreaking treatments to patients. As an innovation-driven and customer-focused company, WuXi AppTec helps our partners improve the productivity of advancing healthcare products through cost-effective and efficient solutions. With industry-leading capabilities such as R&D and manufacturing for small molecule drugs, cell and gene therapies, and testing for medical devices, WuXi AppTec’s open-access platform is enabling more than 4,400 collaborators from over 30 countries to improve the health of those in need – and to realize our vision that "every drug can be made and every disease can be treated." Please visit: http://www.wuxiapptec.com
Forward-Looking Statements
This press release may contain certain “forward-looking statements” which are not historical facts, but instead are predictions about future events based on our beliefs as well as assumptions made by and information currently available to our management. Although we believe that our predictions are reasonable, future events are inherently uncertain and our forward-looking statements may turn out to be incorrect. Our forward-looking statements are subject to risks relating to, among other things, the ability of our service offerings to compete effectively, our ability to meet timelines for the expansion of our service offerings, our ability to protect our clients’ intellectual property, unforeseeable international tension, competition, the impact of emergencies and other force majeure. Our forward-looking statements in this press release speak only as of the date on which they are made, and we assume no obligation to update any forward-looking statements except as required by applicable law or listing rules. Accordingly, you are strongly cautioned that reliance on any forward-looking statements involves known and unknown risks and uncertainties. All forward-looking statements contained herein are qualified by reference to the cautionary statements set forth in this section. All information provided in this press release is as of the date of this press release and are based on assumptions that we believe to be reasonable as of this date, and we do not undertake any obligation to update any forward-looking statement, except as required under applicable law.
Use of Non-IFRS and Adjusted Non-IFRS Financial Measures
We provide non-IFRS gross profit and non-IFRS net profit attributable to owners of the Company, which exclude share-based compensation expenses, issuance expenses of convertible bonds, fair value gain or loss from derivative component of convertible bonds, foreign exchange-related gains or losses, amortization of intangible assets acquired in business combinations and goodwill impairment. We also provide adjusted non-IFRS net profit attributable to owners of the Company and earnings per share, which further exclude realized and unrealized gains or losses from our venture investments and joint ventures. Neither is required by, or presented in accordance with IFRS. We believe that the adjusted financial measures used in this press release are useful for understanding and assessing our core business performance and operating trends, and we believe that management and investors may benefit from referring to these adjusted financial measures in assessing our financial performance by eliminating the impact of certain unusual, non-recurring, non-cash and non-operating items that we do not consider indicative of the performance of our core business. Such adjusted non-IFRS net profit attributable to owners of the Company, the management of the Company believes, is widely accepted and adopted in the industry the Company is operating in. However, the presentation of these adjusted non-IFRS financial measures is not intended to be considered in isolation or as a substitute for the financial information prepared and presented in accordance with IFRS. You should not view adjusted results on a stand-alone basis or as a substitute for results under IFRS, or as being comparable to results reported or forecasted by other companies.
For more information, please contact:
Mr. Bob Shen (For investors)
IR Director
Email: shen_yunbo@wuxiapptec.com
Mr. Davy Wu (For media)
PR Director
Email: davy_wu@wuxiapptec.com
