WuXi AppTec Reports Record Year-to-Date Results in 2022, Third Quarter Revenue over RMB10 Billion
2022/10/26
Revenue Up 71.9% Year-Over-Year to RMB28,395 Million
Net Profit Attributable to Owners of the Company Up 107.1% Year-Over-Year to RMB7,378 Million
Diluted Earnings Per Share (EPS) Up 90.1% Year-Over-Year to RMB2.30
Adjusted Non-IFRS Net Profit Attributable to Owners of the Company Up 77.9% Year-Over-Year to RMB6,772 Million
Adjusted Non-IFRS Diluted EPS Up 76.2% Year-Over-Year to RMB2.29[1]
Operating Cash Flow Up 142.1% Year-Over-Year to RMB7,431 Million
(SHANGHAI, October 26, 2022) — WuXi AppTec (stock code: 603259.SH / 2359.HK), a global company that provides a broad portfolio of R&D and manufacturing services that enable companies in the pharmaceutical, biotech and medical device industries to advance discoveries and deliver groundbreaking treatments to patients, is pleased to announce its financial results for the third quarter and nine months ended September 30, 2022 (“Reporting Period”).
This release provides a summary of the results and is not intended to be a comprehensive report. For additional information, please refer to the 2022 Third Quarterly Report and other relevant announcements published on the websites of the Shanghai Stock Exchange (www.sse.com.cn) and the Stock Exchange of Hong Kong (www.hkexnews.hk), and the designated media for dissemination of the relevant information. Investors are advised to exercise caution and be aware of the investment risks in trading Company shares.
All financial information disclosed in this press release is prepared based on International Financial Reporting Standards (IFRS), in currency of RMB.
The 2022 Third Quarterly Report of the Company has not been audited.
Third Quarter 2022 Financial Highlights
Revenue grew 77.8% year-over-year to RMB10,638 million. This is primarily attributable to the Company’s continuing strong execution of our unique Contract Research, Development and Manufacturing Organization (CRDMO) business model to achieve synergy and strong growth:
-WuXi Chemistry revenue grew 114.8% to RMB7,843 million and adjusted non-IFRS gross profit grew 117.0% to RMB3,262 million, with a gross profit margin of 41.6%.
-WuXi Testing revenue grew 28.1% to RMB1,572 million and adjusted non-IFRS gross profit grew 54.3% to RMB612 million, with a gross profit margin of 38.9%.
-WuXi Biology revenue grew 36.6% to RMB689 million and adjusted non-IFRS gross profit grew 12.3% to RMB288 million, with a gross profit margin of 41.8%.
-WuXi ATU revenue grew 8.4% to RMB306 million and adjusted non-IFRS gross profit was RMB(17) million, with a gross profit margin of -5.5%
-WuXi DDSU revenue declined 30.3% to RMB217 million and adjusted non-IFRS gross profit declined 58.5% to RMB49 million, with a gross profit margin of 22.7%.
[1] In first three-quarters of 2021 and 2022, WuXi AppTec had a fully-diluted weighted average share count of 2,931,763,230 and 2,951,099,205 ordinary shares, respectively.
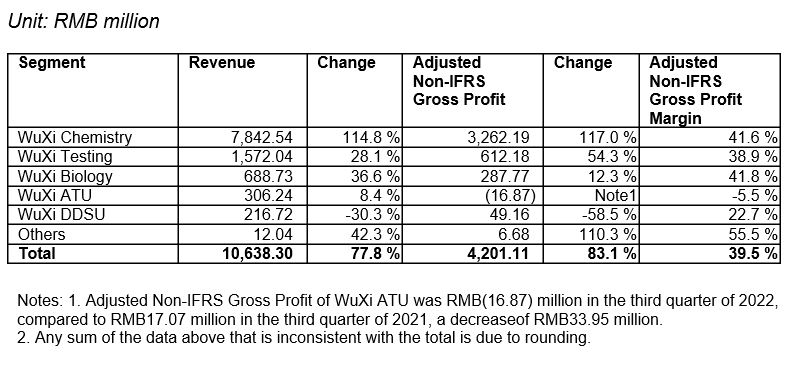
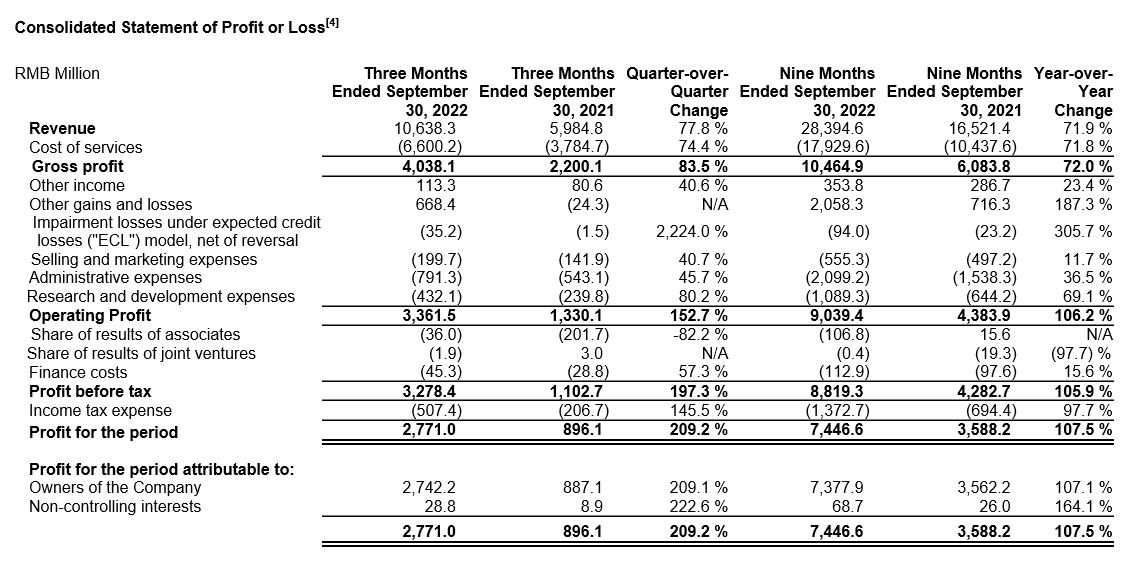
Ø IFRS gross profit increased 83.5% year-over-year to RMB4,038 million. Gross profit margin was 38.0%.[1]
Ø Adjusted Non-IFRS gross profit increased 83.1% year-over-year to RMB4,201 million. Adjusted Non-IFRS gross margin was 39.5%.
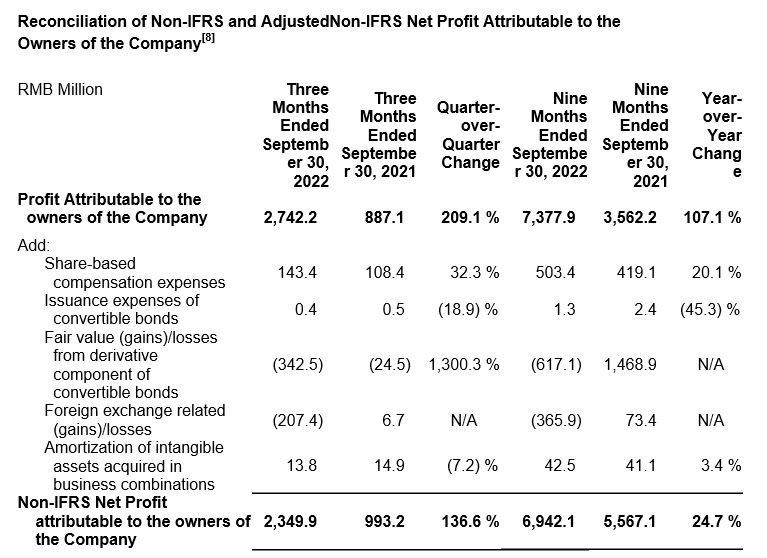
Ø Net profit attributable to owners of the Company increased 209.1% year-over-year to RMB2,742 million.
Ø Adjusted non-IFRS net profit attributable to owners of the Company increased 81.9% year-over-year to RMB2,471 million.
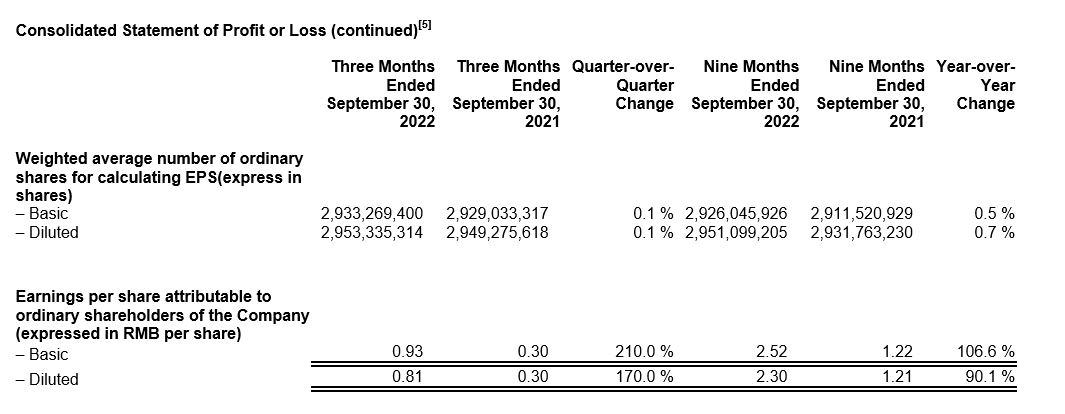
Ø Diluted EPS increased 170.0% year-over-year to RMB0.81, while adjusted diluted non-IFRS EPS increased by 82.6% year-over-year to RMB0.84.
Year-to-Date 2022 Financial Highlights
Revenue grew 71.9% year-over-year to RMB28,395 million for the nine months ended September 30, 2022 (“Reporting Period”):
-WuXi Chemistry revenue grew 106.6% to RMB20,817 million and adjusted non-IFRS gross profit grew 102.4% to RMB8,576 million, with a gross profit margin of 41.2%.
-WuXi Testing revenue grew 25.3% to RMB4,177 million and adjusted non-IFRS gross profit grew 36.0% to RMB1,539 million, with a gross profit margin of 36.8%.
-WuXi Biology revenue grew 24.9% to RMB1,779 million and adjusted non-IFRS gross profit grew 22.8% to RMB732 million, with a gross profit margin of 41.1%.
-WuXi ATU revenue grew 25.2% to RMB922 million and adjusted non-IFRS gross profit was RMB(60) million, with a gross profit margin of -6.5%.
-WuXi DDSU revenue declined 27.9% to RMB672 million and adjusted non-IFRS gross profit declined 54.2% to RMB189 million, with a gross profit margin of 28.1%.
[2] If prepared under Accounting Standard for Business Enterprises of PRC, the gross profit grew 85.4% year-over-year to RMB 4,104 million. Gross profit margin was 38.6%.
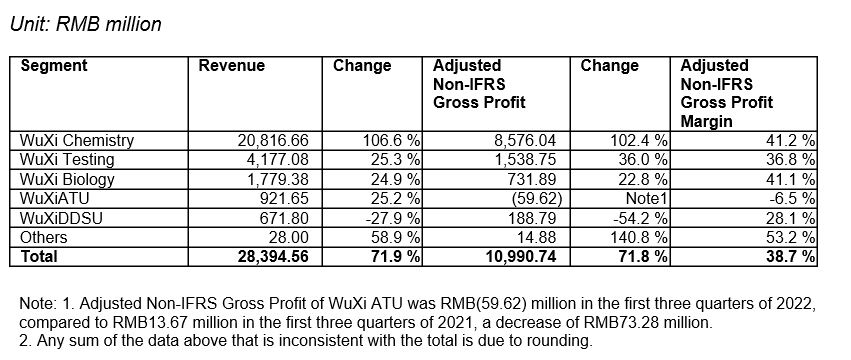
Ø IFRS gross profit increased 72.0% year-over-year to RMB10,465 million. Gross profit margin was 36.9%.[1]
Ø Adjusted Non-IFRS gross profit increased 71.8% year-over-year to RMB10,991 million. Adjusted Non-IFRS gross margin was 38.7%.
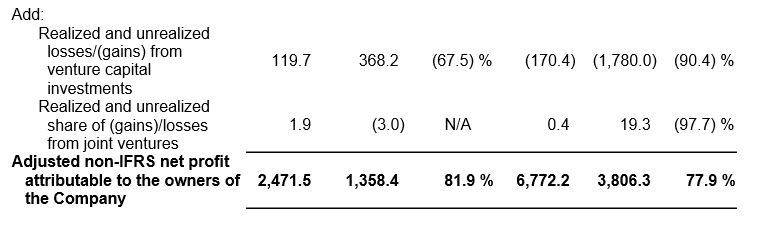
Ø Net profit attributable to owners of the Company increased 107.1% year-over-year to RMB7,378 million.
Ø Adjusted non-IFRS net profit attributable to owners of the Company increased 77.9% year-over-year to RMB6,772 million.
Ø Diluted EPS increased 90.1% year-over-year to RMB2.30, while adjusted diluted non-IFRS EPS increased by 76.2% year-over-year to RMB2.29.
Ø Operating cash flow increased 142.1% year-over-year to RMB7,431 million.
[3] If prepared under Accounting Standard for Business Enterprises of PRC, the gross profit grew 72.5% year-over-year to RMB10,541 million. Gross profit margin was 37.1%
Year-to-Date 2022 Business Operation Highlights
Ø During the first nine months of 2022, demand for our services was strong and we grew our customer base to more than 5,900 active accounts by adding over 1,000 new customers. We continued to optimize our cross-platform synergies to better serve our customers worldwide, strengthen our unique competitive advantage as a fully integrated Contract Research, Development and Manufacturing Organization (CRDMO) and Contract Testing, Development and Manufacturing Organization (CTDMO) platform, and provide one-stop services for our clients from discovery to development and manufacturing. Revenue growth was demonstrated across our expanding global customer base:
-Revenue from US-based customers grew 110% to RMB18,991 million; revenue from Europe-based customers grew 21% to RMB2,897 million; revenue from China-based customers grew 30% to RMB5,318 million; and revenue from other regions grew 22% to RMB1,188 million.
-We continued to expand our customer base and retain existing clients. During the Reporting Period, revenue from existing clients grew 80% to RMB27,546 million and new clients contributed RMB849 million in revenue.
-During the Reporting Period, revenue from the top 20 global pharmaceutical companies grew 175% to RMB12,918 million; revenue generated from all other customers grew 31% to RMB15,477 million.
-Our unique positioning across the pharmaceutical development value chain drove our “follow-the-customer” and “follow-the-molecule” strategies and enhanced synergies across our business segments. Customers using services from multiple business units contributed RMB25,915 million in revenue, growing 83% year-over-year.
Ø WuXi Chemistry: CRDMO integrated business model doubles revenue
-Revenue grew 106.6% to RMB20,817 million and adjusted non-IFRS gross profit grew 102.4% to RMB8,576 million, with a gross margin of 41.2%. Excluding COVID-19 commercial projects, WuXi Chemistry revenue grew 38.3%. Revenue from new modalities-related services grew 192.5%.
-Revenue from small molecule discovery services (“R”) grew 36.2% to RMB5,394 million.
i. Our industry-leading small molecule drug discovery platform delivered more than 379,000 custom synthesized compounds to our clients in the past twelve months. Through our small molecule discovery services, we enabled our customers to accelerate their research while generating opportunities for our downstream business units. As part of our “follow-the-customer” and “follow-the-molecule” strategies, we established trusted partnerships with our global customers, supporting the rapid and sustainable growth of our CRDMO business.
ii. We continued executing our “long-tail” strategy, and those customers continue to have strong demands for our discovery services in small molecules, oligonucleotides and peptides.
-Revenue from our small molecule development and manufacturing (D&M) service grew 152.2% to RMB15,423 million.
i. During the Reporting Period, the Company added 692 new molecules to our D&M pipeline, including 1 new molecule at the commercialization stage. To date, our pipeline consists of 2,123 molecules, including 44 in commercial stage, 53 in phase III, 288 in phase II and 1,738 in phase I and pre-clinical stages.
ii. D&M services for new modalities is also gaining strong momentum. During the Reporting Period, the number of oligonucleotide and peptide D&M clients increased 98% to 97 customers, and the number of oligonucleotide and peptide molecules increased 101% to 155 molecules. Revenue from oligonucleotide and peptide D&M grew 406% to RMB1,101 million.
-We continued our capacity construction of WuXi Chemistry to meet customer demands. During the Reporting Period, we began operations in part of our Changzhou Phase 3 facility, including a new R&D center and 5 plants. We also announced the opening of a new large-scale oligonucleotide and peptide manufacturing facility in July 2022. The new facility underscores our ongoing commitment to enhancing our capacity and capabilities to meet the fast-growing customer needs for oligonucleotide and peptide therapeutics development and manufacturing worldwide. In September 2022, we opened a new sterile lipid nanoparticle (LNP) formulation development and manufacturing facility as well as our first high potency oral drug product manufacturing facility at our Wuxi campus, which further enhances our global integrated CRDMO platform capability. In addition, we opened a new campus in Wuhan, continuously expanding our capacity for drug discovery service. In the third quarter of 2022, we also announced the construction plan for a new site in Singapore. We also held a groundbreaking ceremony for our new pharmaceutical manufacturing campus in Middletown, Delaware, USA to serve our global customers’ future needs.
Ø WuXi Testing: strong growth in lab testing services
-Revenue from WuXi Testing grew 25.3% to RMB4,177 million and adjusted non-IFRS gross profit grew 36.0% to RMB1,539 million, with a gross margin of 36.8%.
-Revenue from lab testing services grew 35.2% year-over-year to RMB3,042 million.
i. The Company provides a full range of laboratory testing services for our customers, including drug metabolism and pharmacokinetics (DMPK), toxicology, and bioanalysis for drug development testing as well as medical device testing. We leveraged our integrated WuXi AppTec Investigational New Drug (WIND) program to generate preclinical data and prepare global regulatory submissions of investigational new drug (IND) packages, expediting the IND application process for many customers worldwide. We signed 89 WIND service packages with customers from January to September 2022.
ii. Drug safety evaluation services achieved strong revenue growth of 49% year-over-year. We maintained our industry-leading position in Asia Pacific for drug safety evaluation services that meet global regulatory requirements.
iii. Our largely US-based medical device testing business has turned around and grew 30% year-over-year.
iv. On the capacity side, a 55,000 square meters lab and animal facilities were under construction for mid-2023 delivery.
-Our clinical CRO & SMO (Site Management Organization) reached a revenue of RMB1,135 million, slowed to a growth of 4.6%, due in part to the ongoing impacts of COVID-19 in many cities across China this year. Meanwhile, our clinical business has rebounded by approximately 95%.
i. For clinical CRO, despite the challenges this year, the Company provided services to approximately 190 projects, enabling our customers to obtain 10 IND approvals.
ii. For SMO, the Company maintained No.1 leadership position in China and continued to grow. Our SMO business maintained more than 4,700 staff across around 150 cities in China, providing services at more than 1,000 hospitals. During the Reporting Period, revenue from our SMO business grew 20.0% and backlog grew 34.9%. From January to September 2022, our SMO business supported 23 new drug approvals for clients.
Ø WuXi Biology: new modalities-related biology services drive growth
-Revenue from WuXi Biology grew 24.9% to RMB1,779 million and adjusted non-IFRS gross profit grew 22.8% to RMB732 million, with a gross margin of 41.1%. Although revenue growth in the second quarter was temporarily impacted by the ongoing effects of the COVID-19 epidemic in Shanghai, business recovered in the third quarter due to the dedication of our employees.
-The Company has one of the largest discovery biology enabling platform, with more than 2,800 experienced scientists who provide comprehensive biology services covering all major stages and therapeutic areas of drug discovery. The Company has established 3 centers of excellence for non-alcoholic steatohepatitis (NASH), anti-viral, neuroscience and aging.
-Both cancer discovery service, rare and immune disease service continued to grow, providing customers with integrated high-quality services from target discovery to clinical biomarker detection.
-The Company has a leading DNA Encoded Library (DEL) and compound generation platform, providing these services for more than 1,300 customers. A customer advanced a project into clinical stage based on the hits generated using our DEL screening technology. This is the first reported clinical candidate from our DEL hits, which is also an important validation of our platform.
-The Company continues to build its biology capabilities for new modalities, including target protein degradation, nucleic acid-based and conjugated modalities, oncolytic virus, vector platform, and novel drug delivery vehicles. During the Reporting Period, WuXi Biology revenue from new modalities and large molecules grew 76%. Its revenue contribution grew to 20.5% year-to-date 2022 from 14.6% in 2021, suggesting that new modalities-related biology services have become an increasingly important driver of growth.
Ø WuXi ATU: CTDMO business model drives growth
-Revenue from WuXi ATU grew 25.2% year over year to RMB922 million and adjusted non-IFRS gross profit was RMB(60) million, with a gross margin of -6.5%. Revenue from Testing service grew 44%, and revenue from Development service grew 54%. Gross profit declined largely due to under-utilized capacities of the newly built Shanghai Lingang site in China.
-During the Reporting Period, the Company focused on improving our CTDMO integrated enabling platform and strengthened testing services, capabilities, and capacities. We provided development and manufacturing services for 67 projects, including 50 pre-clinical and Phase I projects, 9 Phase II projects, and 8 Phase III projects (4 projects are in BLA preparation stage). Up to date, we have supported two BLA filings on behalf of our clients, including one for tumor-infiltrating lymphocytes (TILs) from an US-based client and one for Lenti-viral vector used for CAR-T product from a China-based client.
-The Company announced the launch of Tetracycline-Enabled Self-Silencing Adenovirus (TESSA™) in March 2022. TESSA™ technology is a revolutionary and novel process for transfection-free, scalable manufacturing of adeno-associated virus (AAV). This process is now scaled to 200L and produces more than 10 times the amount of AAV products produced through traditional plasmid-based manufacturing.
-In August 2022, we announced a Licensing Agreement with Janssen for TESSA™ Technology.
-At the end of the third quarter of 2022, we had 38 TESSA™ evaluation projects with customers, including 6 large biotech/pharm companies.
Ø WuXi DDSU: business evolving to focus on innovative drug discovery services
-Revenue from WuXi DDSU declined 27.9% to RMB672 million and adjusted non-IFRS gross profit declined 54.2% to RMB189 million, with a gross margin of 28.1%. DDSU’s revenue decline was mainly attributed to business evolution that aims to better serve the growing needs of clients in China for novel and innovative products. Mainly focused on the discovery of potential best-in-class molecules and new modalities molecules, the business will set a higher bar for R&D and requires longer development cycle, which will impact the business performance in the current period.
-From January to September 2022, our success-based drug discovery service unit filed INDs for 15 drug candidates and obtained 26 Clinical Trial Applications (CTAs), exceeding the total number of 23 CTAs for the whole year in 2021. As of September 30, 2022, we have cumulatively submitted 159 new chemical entity IND filings with the National Medical Products Administration (NMPA) and obtained 136 CTAs, with 1 project in NDA review stage, 5 projects in Phase III clinical trials, 21 projects in Phase II clinical trials, and 77 projects in Phase I clinical trials. We will begin receiving royalty income once our customers launch these products in the market.
-We are now putting our efforts towards the discovery of potential best-in-class molecules and new modalities molecules for customers. Currently, we support 15 pre-clinical projects for customers in new modalities that include Peptide/Peptide-Drug-Conjugation (PDC), protein degraders and oligonucleotides. Several of these projects are expected to file an IND in late 2022 or early 2023.
Ø The unique CRDMO and CTDMO business model of the Company continue to successfully drive record high financial performance. The business of the Company continued its healthy growth while large scale commercialization production projects generated strong cash flow, the Company decided to terminate the Proposed Issuance of H Shares which obtained the approval by the CSRC on August 17, 2022.
Our commitment to ESG
As an enable of innovation, a trusted partner, and a contributor to the global healthcare industry, the Company is committed to environmental protection and sustainability, and to be a good global corporate citizenship.
In August 2022, we received a “Strong” management score by Sustainalytics and ranked in the top two percent in the global pharmaceutical industry with a "Low Risk" rating. As of September 23, 2022, we outperformed 98% of our global peers in the 2022 S&P Global Corporate Sustainability Assessment. In addition, we received an "AA" rating for a second consecutive year in the 2022 MSCI ESG Ratings.
By the third quarter in 2022, our carbon emission intensity, energy consumption intensity and water use intensity reduced by 16.8%, 19.8%, and 22.0%, respectively, compared to the same period of 2021.
Management Comment
Dr. Ge Li, Chairman and CEO of WuXi AppTec, said, “We achieved record growth for the first nine months of 2022. Our revenue increased 71.9% year-over-year and our adjusted Non-IFRS net profit attributable to owners of the company increased 77.9% year-over-year. Our third-quarter revenue reached the milestone of over RMB 10 Billion for the first time in our history. WuXi AppTec’s performance during the first three quarter of 2022 demonstrate again the Company’s unique CRDMO and CTDMO business models allow us to better meet growing demands from customers worldwide and continue to drive rapid growth for the company.”
“As COVID-19 continue to create uncertainty for many around the world, we effectively implemented our business continuity plan and leveraged our global capacities and comprehensive capabilities, to ensure we were able to advance our customers important and life-changing work. We remain committed to putting our customers first. The Company will further enhance our capacity and capabilities to support our partners in their efforts to bring groundbreaking therapies to patients around the world. The Company is confident to adjust the revenue growth target for the full year of 2022 from the previously announced 68%-72% to 70%-72%, and remains the 2021-2024 revenue CAGR growth target of no less than 34%, reflecting the Company's confidence in its continuous accelerated growth driven by CRDMO and CTDMO business models.”
“The Company has achieved excellent results in ESG in the third quarter. The Company remains committed to ’doing the right thing and doing it right.’ We will continue to focus on delivering our ESG commitments and ensuring ESG remain a priority across our business operations today and in the future.”

About WuXi AppTec
As a global company with operations across Asia, Europe, and North America, WuXi AppTec provides a broad portfolio of R&D and manufacturing services that enable the global pharmaceutical and healthcare industry to advance discoveries and deliver groundbreaking treatments to patients. Through its unique business models, WuXi AppTec’s integrated, end-to-end services include chemistry drug CRDMO (Contract Research, Development and Manufacturing Organization), biology discovery, preclinical testing and clinical research services, cell and gene therapies CTDMO (Contract Testing, Development and Manufacturing Organization), helping customers improve the productivity of advancing healthcare products through cost-effective and efficient solutions. WuXi AppTec received an AA ESG rating from MSCI in 2022 and its open-access platform is enabling more than 5,900 customers from over 30 countries to improve the health of those in need – and to realize the vision that "every drug can be made and every disease can be treated." Please visit: http://www.wuxiapptec.com
Forward-Looking Statements
This press release may contain certain “forward-looking statements” which are not historical facts, but instead are predictions about future events based on our beliefs as well as assumptions made by and information currently available to our management. Although we believe that our predictions are reasonable, future events are inherently uncertain and our forward-looking statements may turn out to be incorrect. Our forward-looking statements are subject to risks relating to, among other things, the ability of our service offerings to compete effectively, our ability to meet timelines for the expansion of our service offerings, our ability to protect our clients’ intellectual property, unforeseeable international tension, competition, the impact of emergencies and other force majeure. Our forward-looking statements in this press release speak only as of the date on which they are made, and we assume no obligation to update any forward-looking statements except as required by applicable law or listing rules. Accordingly, you are strongly cautioned that reliance on any forward-looking statements involves known and unknown risks and uncertainties. All forward-looking statements contained herein are qualified by reference to the cautionary statements set forth in this section. All information provided in this press release is as of the date of this press release and are based on assumptions that we believe to be reasonable as of this date, and we do not undertake any obligation to update any forward-looking statement, except as required under applicable law.
Use of Non-IFRS and Adjusted Non-IFRS Financial Measures
We provide non-IFRS gross profit and non-IFRS net profit attributable to owners of the Company, which exclude share-based compensation expenses, issuance expenses of convertible bonds, fair value gain or loss from derivative component of convertible bonds, foreign exchange-related gains or losses, amortization of intangible assets acquired in business combinations and goodwill impairment. We also provide adjusted non-IFRS net profit attributable to owners of the Company and earnings per share, which further exclude realized and unrealized gains or losses from our venture capital investments and joint ventures. Neither is required by, or presented in accordance with IFRS.
We believe that the adjusted financial measures used in this press release are useful for understanding and assessing our core business performance and operating trends, and we believe that management and investors may benefit from referring to these adjusted financial measures in assessing our financial performance by eliminating the impact of certain unusual, non-recurring, non-cash and non-operating items that we do not consider indicative of the performance of our core business. Such adjusted non-IFRS net profit attributable to owners of the Company, the management of the Company believes, is widely accepted and adopted in the industry the Company is operating in. However, the presentation of these adjusted non-IFRS financial measures is not intended to be considered in isolation or as a substitute for the financial information prepared and presented in accordance with IFRS. You should not view adjusted results on a stand-alone basis or as a substitute for results under IFRS, or as being comparable to results reported or forecasted by other companies.
For more information, please contact:
Mr. Ruijia Tang (for investors)
IR Director
Email: tang_ruijia@wuxiapptec.com
Media Contact
Email: mediainquiries@wuxiapptec.com
