Redefining the Undruggable: Covalent Therapies Driving a New Era for Small Molecules
2026/01/19
With advances in biopharmaceutical research and development, next-generation covalent drugs have emerged as a major focus across the industry. To date, more than 50 covalent drugs have been approved for clinical use, demonstrating substantial value across a broad range of therapeutic areas. Importantly, these next-generation therapies are advancing toward reversible covalent binding and targeting amino acid residues beyond cysteine, thereby opening new possibilities for addressing previously “undruggable” targets. Leveraging its fully integrated, end-to-end CRDMO enabling platform, WuXi AppTec provides global partners with comprehensive solutions for covalent drug discovery and development. This article highlights WuXi AppTec’s capabilities and strengths in enabling innovation in covalent drug discovery.
Covalent drugs have become a powerful force in modern drug discovery, continuously expanding the boundaries of therapeutic innovation. As early as the last century, aspirin, with its well-recognized anti-inflammatory and analgesic effects, and penicillin, the first antibiotic effective against a wide range of bacterial infections, demonstrated the immense clinical value of covalent interactions. Unlike traditional non-covalent inhibitors, covalent drugs form stable chemical bonds with their targets, enabling durable inhibition. This unique mechanism allows for lower dosing requirements and higher target occupancy, underscoring their broad potential in clinical applications.
Based on their binding characteristics, covalent drugs can be classified into irreversible and reversible categories. Irreversible inhibitors, such as penicillin, provide long-lasting suppression, while reversible covalent drugs like bortezomib retain a degree of flexibility, balancing durability with tunability. Over the past decade, covalent drugs have entered a phase of rapid growth. By 2024, more than 50 covalent drugs had been approved for clinical use across a wide range of therapeutic areas, including oncology, infectious diseases, genetic disorders, and cardiovascular conditions. Among them, ibrutinib (a BTK inhibitor) and afatinib (an EGFR inhibitor) were the first rationally designed covalent inhibitors. Their success not only reaffirmed the clinical value of covalent bonding but also established a key development strategy: introducing covalent warheads into reversible scaffolds to significantly enhance potency and selectivity.

Beyond this strategy, the approval of KRAS G12C inhibitors sotorasib and adagrasib represents a landmark in the clinical application of a new paradigm in covalent drug discovery. Confronting KRAS—a target long regarded as “undruggable”—researchers adopted a direct-screening strategy to identify small molecules capable of rapidly forming covalent bonds with the mutant cysteine residue. Unlike traditional approaches that relied on modifying existing non-covalent scaffolds, this method focused directly on the mutation site to achieve covalent binding and the optimization of molecules against a new target. This signaled a forward-looking shift in covalent drug discovery from conventional molecular modification to innovative, target-driven design.
At the same time, next-generation covalent drugs are progressing rapidly toward improved selectivity. In parallel, they are being developed to address an increasingly broad spectrum of therapeutic targets. While earlier strategies largely focused on cysteine residues, new approaches are expanding to lysine, serine, threonine, and tyrosine. By incorporating novel covalent warhead chemistries, these strategies aim to achieve higher specificity with reduced off-target risk. This innovative path not only promises to improve the safety and efficacy of covalent therapies but also offers new solutions for protein targets once deemed intractable—ultimately delivering tangible clinical benefits to patients across a wide range of diseases.
To help global partners accelerate the development of next-generation covalent drugs, from early discovery through IND-enabling studies, WuXi AppTec has established an integrated platform. By combining three complementary, state-of-the-art technologies—covalent DNA-Encoded Library (cDEL), covalent Fragment-Based Drug Discovery (cFBDD), and covalent High-Throughput Screening (cHTS)—the platform provides a comprehensive solution to advance covalent drug discovery.
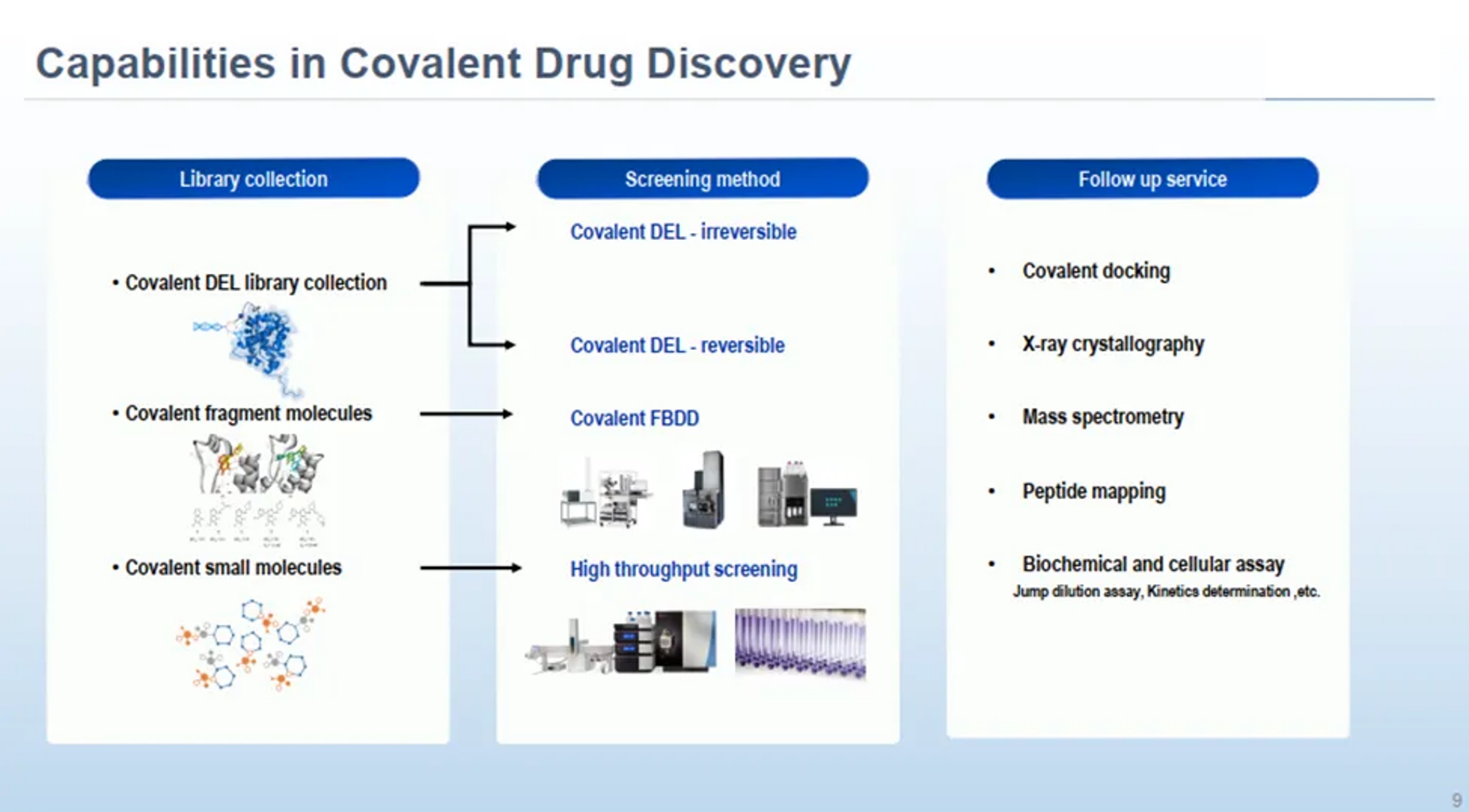
cDEL is a powerful screening technology that enables the efficient exploration of vast chemical libraries, greatly increasing the likelihood of identifying covalent binders with diverse warheads. Leveraging DNA tags as molecular barcodes, the platform allows rapid and cost-effective deconvolution of hits while requiring only minimal quantities of protein and compound. This makes it particularly valuable in the early discovery stage, where broad chemical diversity is essential. Through tailored selection strategies, WuXi AppTec’s cDEL platform uncovers both reversible and irreversible covalent hits, which are further validated through decoding, resynthesis, and rigorous on-DNA and off-DNA assays. For example, for irreversible covalent screening, customers may access DELink Pro, a customizable screening solution leveraging a broad and diverse compound library with extensive warhead coverage, or DELink Lite, a streamlined platform optimized for ease of use.
WuXi AppTec’s cFBDD platform builds on a rigorously curated library of more than 2,600 structurally diverse fragments, each designed under the “rule of three (Ro3)” to ensure optimal physical properties and chemical diversity. (Ro3 holds that drug fragments should be ≤300 Da, cLogP ≤3, and carry no more than 3 hydrogen bond donors or acceptors to remain small, soluble, and readily optimized.) By focusing on small, low-complexity molecules, the platform efficiently probes binding sites and, through the incorporation of electrophilic fragments, identifies covalent hits that serve as highly effective starting points for optimization. Leveraging high-throughput mass spectrometry together with structural biology methods such as X-ray crystallography and NMR, cFBDD provides atomic-level insights into fragment–protein interactions, enabling rational design and systematic growth of covalent inhibitors. The fragment library is free from reactivity and stability issues, remains synthetically accessible, and delivers high-quality data with speed and precision. The combination of structural precision, rapid turnaround, and efficient hit-to-lead progression makes cFBDD a powerful approach for advancing novel covalent leads, particularly against the most challenging or traditionally “undruggable” targets.
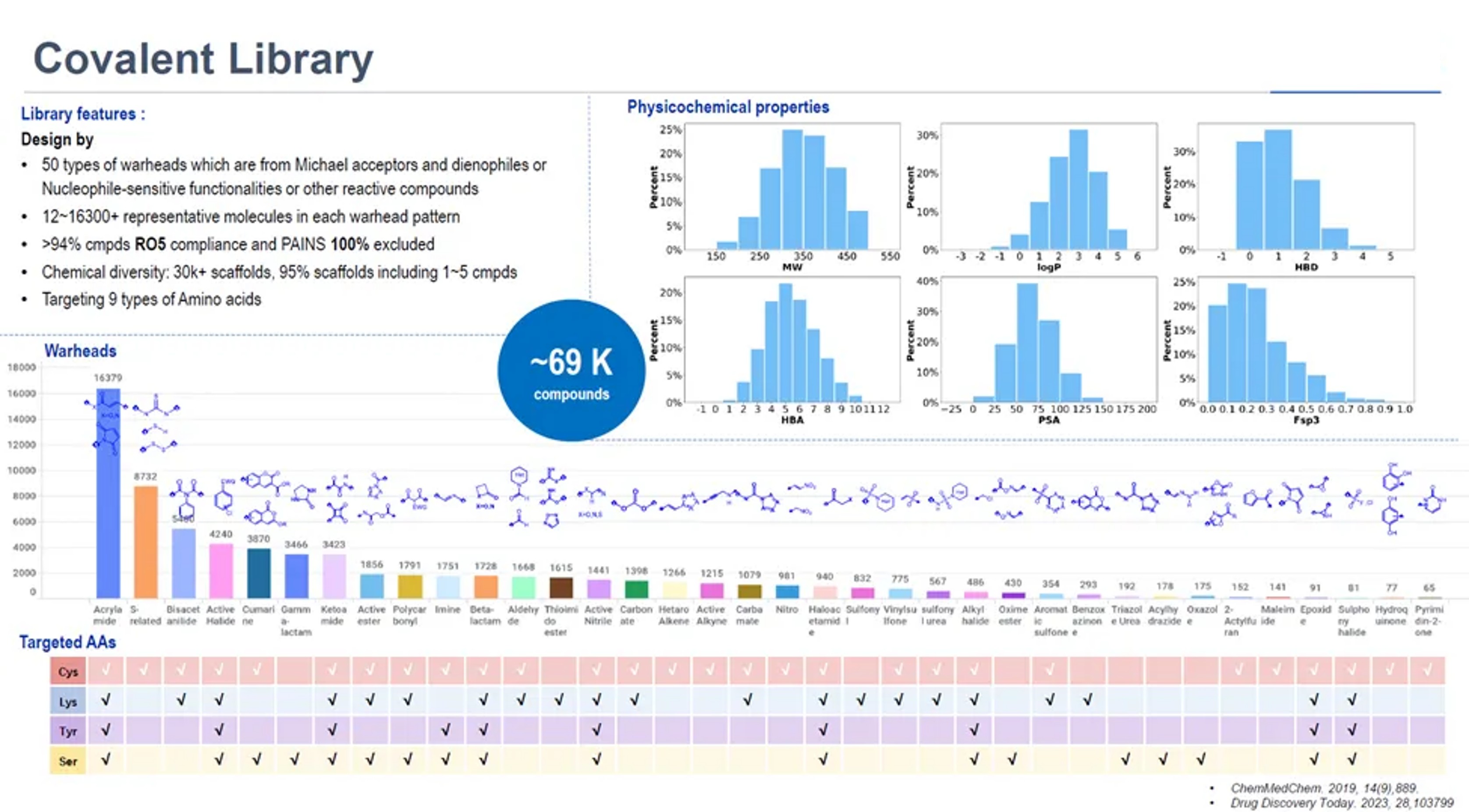
The cHTS platform from WuXi AppTec is a platform that incorporates various screening strategies. The covalent HTS library has approximately 69,000 covalent molecules featuring more than 50 distinct warhead chemotypes across a range of reactivities. Our library is purpose-built to engage with nine different amino acid residues, extending covalent drug discovery beyond cysteine to include serine-, lysine-, and other residue-focused libraries. Because these alternative residues are more abundant in proteins and exhibit environment-specific reactivity, the platform provides access to previously undruggable proteins while improving selectivity and reducing off-target interactions.
The cHTS platform provides different screening methods. The functional-assay-based high-throughput screening, as one of the most established approaches in covalent drug discovery, can test extensive collections of electrophile-containing molecules directly in enzymatic or cellular systems; Mass-Spectrometry-based high-throughput screening can test the molecular weight shifter upon covalent drug binding. These enable rapid generation of functional readouts or mass readouts that confirm actual inhibitory or binding activity, ensuring identification of hits that possess favorable properties for further optimization and clinical development. The cHTS platform can also provide a wide variety of assay for orthogonal hit confirmation, which can transfer the screening hits to confirmed hits. By integrating High-Throughput Chemistry (HTC), the cHTS platform offers a D2B solution that unites fast synthesis and quick-turnaround testing to speed hit expansion. More than a screening engine, cHTS supports the full workflow from Hit ID through Hit-to-Lead.
Beyond functioning as inhibitors, covalent drugs are demonstrating increasingly broad potential in emerging therapeutic approaches, particularly in the field of induced proximity. For example, the discovery of covalent recruiters for E3 ubiquitin ligases has the potential to expand the scope of targeted protein degradation (TPD). At the same time, the identification of covalent recruiters for OTUB1 has led to the development of the deubiquitinase-targeting chimera (DUBTAC) platform, which can stabilize mutant CFTR proteins in patients with cystic fibrosis (CF), offering a new therapeutic direction for this severe genetic disease. Therefore, the value of covalent molecules extends far beyond inhibition, gradually evolving into multidimensional therapeutic strategies that open broader possibilities for innovative drug discovery.
Complementing its advanced screening capabilities, WuXi AppTec offers a comprehensive suite of early discovery technology platforms—including biophysical, biochemical, and cellular assays—to support global partners in advancing covalent drug development. Backed by an integrated, end-to-end CRDMO enabling platform, WuXi AppTec is committed to accelerating the development of transformative therapies that deliver meaningful benefits to patients worldwide—fulfilling its vision that “Every drug can be made and every disease can be treated.”
